Last updated on April 20th, 2021 at 08:23 pm
Fasoracetam is a relatively new racetam that’s actively being developed as a potential pharmaceutical drug. The research so far suggests it may have nootropic properties. Specifically, fasoracetam has been shown to improve ADHD through a novel mechanism involving glutamate rather than dopamine and noradrenaline.
At a Glance
Research suggests fasoracetam may be helpful for:
- ADHD – Clinical trials have shown it can significantly decrease symptoms of ADHD
- Memory – It’s been shown to improve memory and learning in rats. In addition, it can prevent amnesia
- Depression – One study found fasoracetam has significant antidepressant effects in rats
Fasoracetam has been shown to affect:
- Glutamate – Fasoracetam increases glutamate activity in human and animal studies, which is one of the mechanisms it’s thought it reduces ADHD
- GABA – GABAB receptors are upregulated by fasoracetam. Notably, these receptors can increase dopaminergic activity
- Acetylcholine – Levels of acetylcholine appear to increase via 2 mechanisms: higher synthesis and increased release
| Overview | |
|---|---|
| Also Known As | NS-105, NFC-1, (+)-5-oxo-d-prolinepiperidinamide monohydrate, AEVI-001, LAM 105, MDGN-001 |
| Type | Racetam |
| Synergistic with |
|
| Does not go well with |
|
| Typical route | Orally |
| Typical dose | 50 mg twice a day |
| Typically taken | Chronically |
| Half-life | 4-7 hours in humans[12] |
| Where to Buy | Science.bio – Bulk powder Pure Nootropics – Capsules |
What is Fasoracetam
Fasoracetam is one of the newest drug of the racetam family. It was originally developed for vascular dementia, but dropped after achieving poor results in clinical trials.
However, it was picked up again after showing promise in treating ADHD. In fact, it was approved by the FDA’s Investigational New Drug (IND) program in 2015, specifically for ADHD. Since then, a total of 6 studies on fasoracetam’s effect on ADHD have been completed.
Because fasoracetam is not a stimulant, it can be a novel treatment for ADHD. In other words, it might be a viable alternative to Adderall and Ritalin.
Moreover, it’s also being investigated as a potential treatment in Autism Spectrum Disorders (ASD).[13]
Though only a few studies have been done in humans, fasoracetam appears to have nootropic properties. However, more research is needed to confirm its safety and efficacy. As such, it’s safer to brand fasoracetam as a research chemical than a true nootropic.
Benefits of Fasoracetam
From the research so far, it appears that Fasoracetam has 3 major benefits.
May Improve ADHD
This is probably fasoracetam’s most significant benefit and the reason why a pharmaceutical company is trying to get it FDA approved.
Put briefly, fasoracetam has been shown to significantly decrease symptoms of ADHD in multiple studies.[1] However, this was mainly in children and with a specific glutamate mutation. But it doesn’t necessarily mean that people without this mutation can’t benefit from it.
Furthermore, its effects got stronger with time. In contrast, typical ADHD medications like stimulants tend to lose some of their effects with time.
So it’s possible that fasoracetam may be a long-term treatment for ADHD.
May Improve Memory and Learning
It appears that fasoracetam may improve memory and prevent memory impairments. This is based on 2 findings.
The first showed that fasoracetam increases acetylcholine levels. In particular, this neurotransmitter is very important in memory and learning as well as cognition in general.
Because of this, it was shown to effectively blocked amnesia in rats with poor choline function. Similarly, the second showed it restored memory impairments caused by the drug Baclofen. However, in this case it did so by blocking Baclofen’s effects on the GABA system.[3]
May be an Antidepressant
At least 1 study has found it to be promising for depression. In particular, the study compared fasoracetam to the potent antidepressant desipramine.[2]
Specifically, it’s a tricyclic antidepressant (TCA) that mainly inhibits norepinephrine reuptake and to a lesser extent, serotonin. Notably, fasoracetam was as strong as desipramine in reducing the symptoms of depression.
Thus, it appears to have strong antidepressant effects.
How Fasoracetam Feels
Users taking fasoracetam often report a strong anti-anxiety effect.
In some cases, people have said that it is life changing. In particular, one person has found it to greatly improve verbal fluency, decrease dissociation, and decrease anxiety.[14]
Finally, the effects of fasoracetam can show up in as little as 5-10 minutes and tend to last at least 6 hours or more.
Effects of Fasoracetam
- Significantly decreased anxiety[15]
- Substantially improved memory[14]
- Uplifted mood[14][15]
- Reversal of phenibut tolerance[16]
On the other hand, fasoracetam is really a hit-or-miss kind of nootropic. In other words, it doesn’t work for everyone. In fact, it seems to either work completely or not work at all. This is based on anecdotes online.
Interestingly, the outcome is irrelevant of ADHD status. So fasoracetam’s effects on glutamate are likely not responsible for this. Instead, it appears that its effects on the GABA system are the main reason why people report a decrease in anxiety.
How Fasoracetam Works
Current research has shown that it works via 3 ways:
- Increasing glutamate activity
- Upregulating GABAB receptors
- Increasing acetylcholine release and synthesis
Fasoracetam’s main mechanism of action is increasing the activity of glutamate in the brain.[1] As a result, this is thought to be the reason why it decreases ADHD. However, it’s unclear exactly how it does this.
Indeed, glutamate receptors play an important role in both the cause and treatment of ADHD. But the research thus far isn’t conclusive. That is to say, the exact mechanism as to how glutamate receptors affect ADHD isn’t fully known yet.[18]
Moreover, it also increases the amount of GABAB receptors in the brain.[2] In turn, this gives fasoracetam its antidepressant effects. Furthermore, it explains how it can prevent memory impairments caused by the drug Baclofen, which primarily works via the GABAB receptor.[3]
In addition, it also increases the release and synthesis of acetylcholine.[3] This neurotransmitter is especially important in memory and learning. Because of this, it may explain how fasoracetam improves these factors of cognition, at least in rats.
Finally, fasoracetam does not affect[2]:
- Dopamine
- Noradrenaline
- Adrenaline
- Serotonin
Synergies
Fasoracetam may synergize well with antidepressants and ADHD medications. It appears to have a unique mechanism of action that greatly differs from traditional pharmaceutical drugs. Because of this, it should not interact with them, but rather enhance their effects.
However, this is merely a hypothesis and not confirmed by research.
Additionally, fasoracetam may be able to reduce tolerance to phenibut. Specifically, it upregulates GABAB receptors, which is one of phenibut’s key targets. Moreover, people taking fasoracetam have reported that it does indeed synergize with phenibut.[16]
Side effects
In a small study involving adolescents with ADHD[1], the most common side effects were mild in nature and included:
- Headache – 63.3% of subjects
- Fatigue – 36.7%
- Upper abdominal pain – 26.7%
- Diarrhea – 23.3%
- Irritability – 20%
However, it was not a double-blind study and as such there was no placebo-group to compare the side effects with. The researchers also noted that the side effects were not related to fasoracetam.
Safety
In 6 different clinical trials, fasoracetam did not cause any serious side effects. In general, it was found to be safe and well-tolerated.[1]
Phenibut can be dangerous with fasoracetam
At low doses, fasoracetam may combine well with phenibut to create a synergistic effect. However, higher doses may be dangerous. This is due to the fact that fasoracetam increases the number of GABAB receptors in the brain. In short, it is thought that one of phenibut’s primary actions is via the GABAB. Consequently, the two combined together may result in overstimulation of the receptor which can lead to many side effects.
In fact, there’s a case report about this. A 27 year old male combined 10 grams of Phenibut with an unknown amount of fasoracetam and ended up passing out on the sidewalk. He was taken to a hospital and on his second day there he developed psychosis.[19]
Research
Summary of benefits
| Condition | Effect | Dosage | Subjects |
|---|---|---|---|
| ADHD | Significant decrease in symptoms with stronger effects the longer it was taken | 50 - 500 mg twice a day | Adolescents with ADHD and a gene variant of the glutamate receptor |
| Memory and learning | Improved memory Reversed memory disruptions caused by baclofen | 10 mg/kg (rats) | Rats |
| Depression | Strong decrease comparable to desipramine | 1-100 mg/kg (rats) | Rats |
Attention deficit hyperactivity disorder (ADHD)
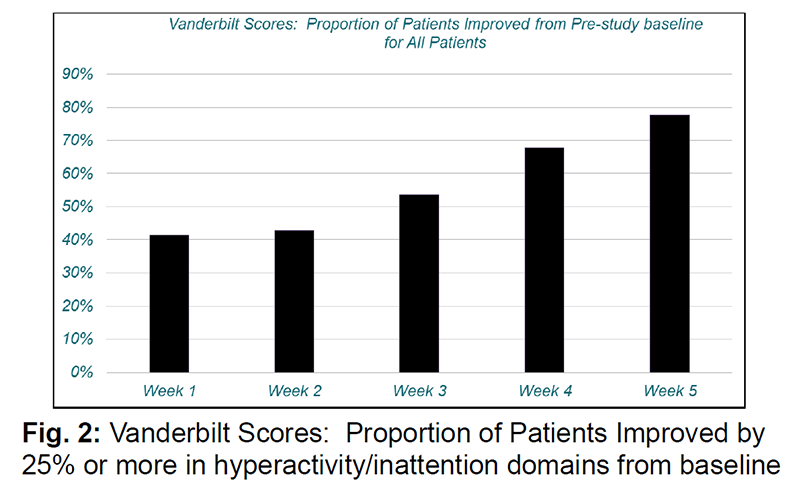
In a study involving 30 adolescents, 29 of which completed it, there was a significant decrease in ADHD symptoms and subsequently Vanderbilt scores, which measures ADHD symptoms. Mean scores at week 1 were 29.1 which decreased to 22.5 by week 5. Doses started at 50 mg twice a day at week 1 and increased weekly to a maximum of 500 mg twice a day at week 5. ADHD symptoms improved at week 2 of the study and continued improving until the end of the study of week 5.[1]
Depression
In rats, fasoracetam demonstrates a potent antidepressant effect comparable to that of desipramine.[2]
Memory and learning
Fasoracetam exhibits antiamnesic properties in rats with cholinergic dysfunction and reverses the memory disruptions caused by baclofen.[3]
Schizophrenia
Schizophrenia is purported to be associated with hypoglutamatergic activity.[4] Fasoracetam was found to reduce ADHD symptoms in adolescents with low glutamate neurotransmission.[1] Extrapolating these results, it seems likely that it may also benefit patients with schizophrenia. However, no studies have been done to assess this hypothesis.
Mechanism of action
Humans
Glutamate
In the ADHD population, there is a significantly higher copy-number variant(duplication of a gene) affecting the metabotropic glutamate receptors genes resulting in hypoglutamatergic activity.[5] Fasoracetam is purported to increase glutamatergic activity and was found to significantly reduce the symptoms of ADHD in adolescents.[1]
Animals and lab studies
| Neurotransmitter | Change | Mechanism | Effects | Subjects |
|---|---|---|---|---|
| Acetylcholine | Increased levels | Increased release and synthesis | Improved memory | Rats with choline dysfunction |
| Dopamine | No change | None | None | Rats |
| Noradrenaline and adrenaline | No change | None | None | Rats |
| GABA | Possible increase in activity | Upregulation of GABAB receptors |
| Rats |
| Glutamate | Increased activity | Unknown | Possible decrease in ADHD symptoms | Rats |
Acetylcholine
In the cerebral cortex and the hippocampus, fasoracetam increases high affinity choline uptake (HACU) which increases acetylcholine synthesis. Additionally, there is an increase in acetylcholine release in the cerebral cortex.[3]
Dopamine and noradrenaline
It appears fasoracetam does not alter dopamine, noradrenaline and adrenaline levels nor does it bind to the beta-adrenoceptors.[2]
GABA
Fasoracetam binds to the GABAB receptor in rats effectively blocking the memory disruptions caused by baclofen. However, it’s unclear whether it activates the receptor (agonist), blocks it (antagonist), or acts in a more complex-manner, i.e. partial-agonist or inverse agonist.[3]
Regardless, it appears to upregulate the GABAB receptors in the cerebral cortex of rats which may contribute to its antidepressant effect.[2]
Glutamate
In the rat brain, fasoracetam inhibits forskolin-induced cAMP formation in the cerebral cortex by stimulating the metabotropic glutamate receptors.[6][7][8]
Serotonin
No change was seen in the serotonin levels of rats. Likewise, it does not appear to bind to the 5-HT2 receptor.[2]
Pharmacokinetics
In humans, the time it takes to fully absorb fasoracetam differs depending on age. For instance, a study compared young adults to seniors with a single 100 mg tablet. In this case, maximum blood concentration was reached after about 1.3 hours in young adults and 2.1 hours in the elderly. [9]
Moreover, the half-life of fasoracetam varies depending on the species:
Furthermore, its bioavailability also differs with different species[10]:
- Rats – 97%
- Dogs – 90%
- Monkeys – 79%
Its bioavailability is quite high for 2 reasons. First, it’s greatly absorbed by the small intestines. And second, it’s not affected by first-pass metabolism. As a result, it has similar oral and intravenous bioavailability.
Moreover, about 90% of fasoracetam is excreted unchanged in the urine of rats and monkeys, with 60% for dogs.[10]
Finally, chronic administration in rats does not affect liver drug-metabolizing enzymes (cytochrome P450s).[11]
References
- Jones, Grace. “Exploratory dose-escalation study of NFC-1 in ADHD adolescents with glutamatergic gene network variants.” 62nd Annual Meeting. AACAP, 2015.
- Shimidzu, Takako et al. “Effect of a Novel Cognition Enhancer NS-105 on Learned Helplessness in Rats: Possible Involvement of GABAB Receptor up-Regulation after Repeated Treatment.” European Journal of Pharmacology, vol. 338, no. 3, 1997, pp. 225–232. doi:10.1016/s0014-2999(97)81925-5.
- Ogasawara, Takashi et al. “Involvement of Cholinergic and GABAergic Systems in the Reversal of Memory Disruption by NS-105, a Cognition Enhancer.” Pharmacology Biochemistry and Behavior, vol. 64, no. 1, 1999, pp. 41–52. doi:10.1016/s0091-3057(99)00108-2.
- Itokawa, M., and T. Yoshikawa. “[Hypoglutamatergic hypothesis of schizophrenia: evidence from genetic studies].” Seishin shinkeigaku zasshi= Psychiatria et neurologia Japonica 105.11 (2002): 1349-1362.
- Elia, Josephine et al. “Genome-Wide Copy Number Variation Study Associates Metabotropic Glutamate Receptor Gene Networks with Attention Deficit Hyperactivity Disorder.” Nature Genetics, vol. 44, no. 1, Apr. 2011, pp. 78–84. doi:10.1038/ng.1013.
- Oka, Michiko et al. “Involvement of Metabotropic Glutamate Receptors in Gi- and Gs-Dependent Modulation of Adenylate Cyclase Activity Induced by a Novel Cognition Enhancer NS-105 in Rat Brain.” Brain Research, vol. 754, no. 1-2, 1997, pp. 121–130. doi:10.1016/s0006-8993(97)00064-4.
- Oka, Michiko et al. “A Novel Cognition Enhancer NS-105 Modulates Adenylate Cyclase Activity through Metabotropic Glutamate Receptors in Primary Neuronal Culture.” Naunyn-Schmiedeberg’s Archives of Pharmacology Naunyn-Schmiedeberg’s Arch Pharmacol, vol. 356, no. 2, 1997, pp. 189–196. doi:10.1007/pl00005040.
- Hirouchi, Masaaki et al. “Role of Metabotropic Glutamate Receptor Subclasses in Modulation of Adenylyl Cyclase Activity by a Nootropic NS-105.” European Journal of Pharmacology, vol. 387, no. 1, 2000, pp. 9–17. doi:10.1016/s0014-2999(99)00785-2.
- Kumagai, Y., et al. “Comparison of pharmacokinetics of NS-105, a novel agent for cerebrovascular disease, in elderly and young subjects.”International journal of clinical pharmacology research 19.1 (1998): 1-8.
- Mukai, Hideya, et al. “Pharmacokinetics of NS-105, a Novel Cognition Enhancer.” Arzneimittelforschung 49.11 (1999): 881-890.
- Mukai H, Sugimoto T, Ago M, Morino A, Takaichi M, Ogawa Y, Seki H, Matsuura C, Esumi Y. Pharmacokinetics of NS-105, a novel cognition enhancer. 2nd communication: distribution and transfer into fetus and milk after single administration, and effects of repeated administration on pharmacokinetics and hepatic drug-metabolizing enzyme activities in rats. Arzneimittelforschung. 1999 Dec;49(12):977-85. PubMed PMID: 10635441.
- Elia, Josephine, et al. “Fasoracetam in adolescents with ADHD and glutamatergic gene network variants disrupting mGluR neurotransmitter signaling.” Nature communications 9.1 (2018): 4.
- A signal finding study of AEVI-001 in Autism Spectrum Disorders (ASD).
- Fasoracetam has changed my life!
- Fasoracetam Experience (> 2 Weeks)
- FASORACETAM – Longecity
- Merchan, Cristian, et al. “Phenibut Overdose in Combination with Fasoracetam: Emerging Drugs of Abuse.” Journal of Clinical Intensive Care and Medicine 1 (2016): 001-004.
- Huang, Xin, et al. “The role of glutamate receptors in attention‐deficit/hyperactivity disorder: From physiology to disease.” American Journal of Medical Genetics Part B: Neuropsychiatric Genetics 180.4 (2019): 272-286.
- Merchan, Cristian, et al. “Phenibut Overdose in Combination with Fasoracetam: Emerging Drugs of Abuse.” Journal of Clinical Intensive Care and Medicine 1 (2016): 001-004.